
Non-Conforming Process – AS9100 has more specific requirements on how to handle non-conformances and how to take action on processes and products, including what corrective actions need to take place if a nonconformance is found.The last seven clauses are now arranged according to the PDCA cycle (Plan, Do, Check, Act).
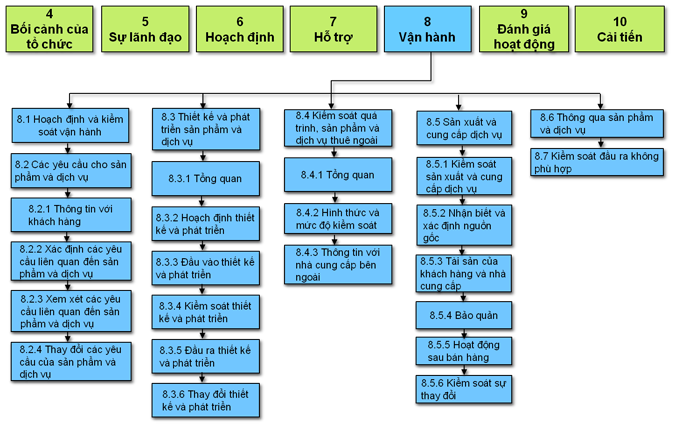
The changes include production processes, as well as required controls on production equipment and support after delivery. ISO 9001:2015: The first three clauses in ISO 9001:2015 are largely the same as those in ISO 9001:2008, but there are considerable differences between ISO 9001:2008 and ISO 9001:2015 from the fourth clause onward. The reason is that this is the biggest difference from one industry to the other.
#Iso 9001:2015 vs iso 13485:2016 matrix manual#
There are many areas that have additional requirements, but the following are the largest differences between the two standards: Correspondence Between ISO 13485:2016 and 21 CFR 820 Regulatory Compliance Associates® Inc., 10411 Corporate Drive, Suite 102, Pleasant Prairie, WI 53158 5:2016 US FDA Quality System Regulation (QSR - 21 CFR 820) The quality manual shall outline the structure of the documentation used in the quality management system. When transitioning from ISO 9001 to AS9100, you will find the additional requirements for AS9100 in italics. What are the additional requirements of AS9100 Rev D compared to ISO 9001:2015?

AS9100 Internal Auditor Training Courses.Simple Steps to AS9100 Certification Follow our proven and manageable step-by-step process for a successful ISO implementation project. In ISO 13485:2016 risk pertains to safety or performance requirements of the medical device or meeting applicable regulatory requirements.What is AS9102 First Article Inspection?.
